Phosphorus in soil
Phosphorus (P) is an essential element for plant growth, but it is also a relatively immobile element in the soil. Fertiliser phosphorus is rapidly immobilised in the soil, with only an estimated 10-20% of applied phosphorus being used by plants in the year following application.
Phosphorus accumulates in the soil in both inorganic and organic forms. Organic phosphorus may account for between 50 and 80% of the phosphorus in soil, with soils in pasture having higher levels. Inorganic phosphorus occurs in different forms: anionic forms adsorb onto soil materials in complexes with iron oxides, aluminium oxides, aluminium silicates and calcium carbonates. Inorganic phosphorus can also precipitate out of solution: in acid soils phosphorus precipitates as iron and aluminium complexes; in alkaline soils it precipitates with calcium.
Phosphorus uptake by plants
Plants can only take up phosphate anions (HPO4 2- and H2PO4 -) that occur in the soil solution. The concentration of these is generally extremely low – less than 10 μM. In contrast, the concentration of phosphorus inside plants is in the millimolar range, i.e. a thousand times greater.
As a result, plants need active, energy-driven transport systems to bring phosphate in against this steep concentration gradient. This is achieved by the use of transporter proteins. Transporting phosphate into the cell reduces the pH in the cytoplasm and to compensate for this the cell ejects hydrogen ions via a proton pump, a process that uses energy (see Figure 1).
Plants have two types of phosphate transporter proteins embedded in their cell membranes and these each operate under specific conditions (see Figure 2).
The high-affinity transporters tend to dominate when phosphate concentrations are low. As they have a high affinity for phosphate, they enable the plant to scavenge effectivelywhen phosphate is scarce.
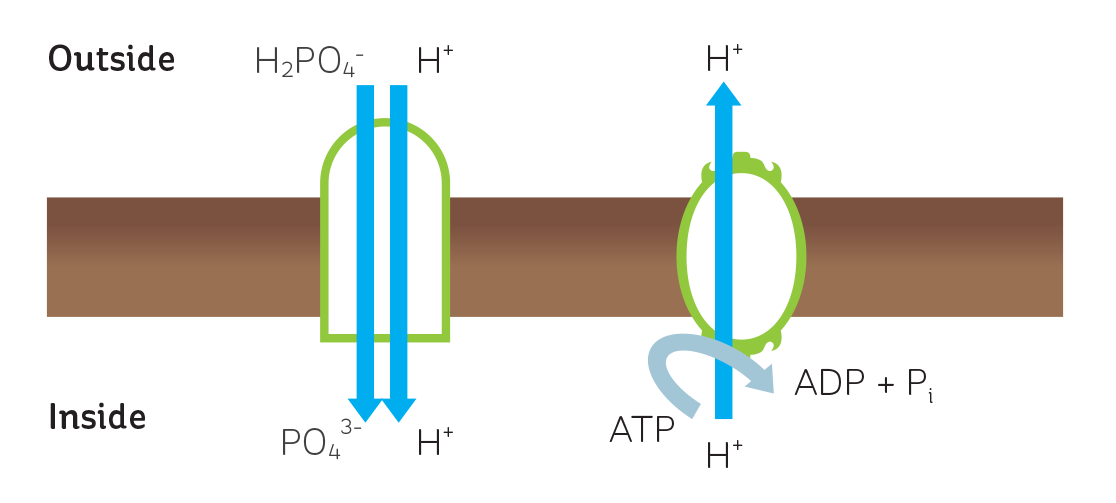
FIGURE 1
A model depicting the mechanism of phosphate transport into cells in plant roots
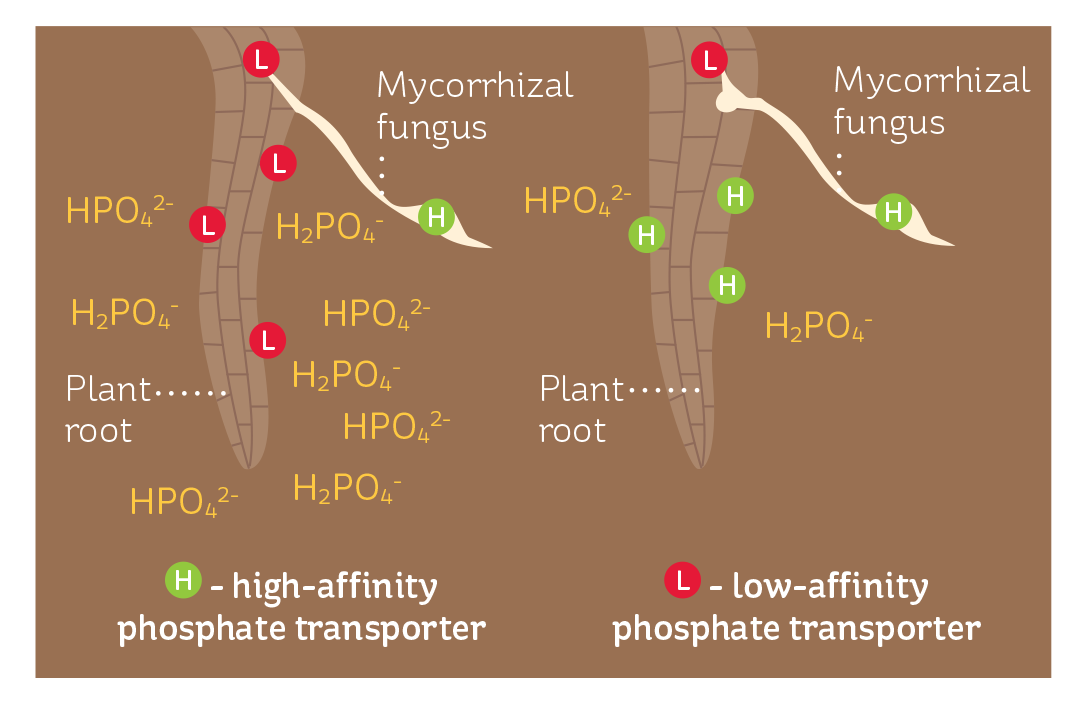
FIGURE 2
When soil phosphate levels are high, plants express lowaffinity transporters; as levels fall, the plant produces highaffinity transporters to more effectively capture the available phosphate shoot concentrations are sufficient. There is also evidence that their expression is suppressed when there is a deficiency of a second nutrient.
The low-affinity transporters are thought to be present all the time. These transporters are also likely to be involved in the transport of phosphate between cells. Regardless of whether they are high or low affinity, phosphate transporter proteins do not remain constantly in the membrane. Rather, they are regularly turned over in response to changes in the presence of inorganic phosphate. This mechanism is thought to prevent plants acquiring toxic levels of phosphorus.
Once phosphate has entered cells it may be used directly or it may be stored in vacuoles for future use. This storage facility is essential for plants to maintain an even supply of phosphorus to cells. Studies have shown that when plants are grown under conditions of phosphate sufficiency, between 85 and 95% of the cellular phosphorus is found in the vacuoles. Increasing the amount of phosphate in the environment does not increase the amount of phosphorus in the cytoplasm, but it does increase the amount of phosphorus stored in the vacuoles.
Enhancing phosphate uptake
Plants have developed a number of other strategies for ensuring adequate supply of phosphorus. When phosphate is in short supply, plants respond by modifying their root morphology and by the preferential growth of roots instead of shoots. They may also secrete compounds that improve phosphate availability.
The following are examples of strategies used by various plants to enhance phosphate availability:
- Preferential growth of roots instead of shoots results in a greater surface area of root matter through which phosphate can be acquired
- Increased branching of roots increases the soil area explored by the roots
- Increased growth of root hairs increases the surface area of the plant’s root system
- Production of the plant hormones auxin and ethylene may influence plant root morphology under low phosphate conditions
- Release of organic acids such as malate, citrate and oxalate into the rhizosphere may help to solubilise phosphate and so increase availability
- Some plants release phosphatases, enzymes that can degrade organic phosphorus and so make it available for plant uptake.
Role of mycorrhizal fungi
Mycorrhizae are fungi that form a close and unique relationship with higher plants, infecting the roots to the ultimate benefit of both parties. The fungi grow in between cells of plant roots and also enter some cells, where they form structures known as arbuscles (which look a bit like little trees growing inside the cell – see Figure 3). The arbuscles are thought to be the site of nutrient transfer between plant and fungus.
Mycorrhizal fungi grow out from the plant root into the soil and so they act as a vast extension of the plant’s root system and enable a much greater area of soil to be accessed, soincreasing the amount of nutrients available to the plant (see Figure 4).
The mycorrhizae take up nutrients from the soil for their own growth and transfer some of these to the plant. In exchange, they receive carbon-based compounds from the plant.
Like plants, mycorrhizal fungi also move phosphate through both high-affinity and low-affinity phosphate transporters. In these fungi, the high-affinity transporter takes up the phosphate from the soil, while the low-affinity transporter delivers it from the fungus to the plant cells. When a plant becomes infected with a mycorrhizal fungus, its own phosphate uptake mechanism is down-regulated, or suppressed, so the vast majority of phosphate entering the cell will be via the mycorrhizal fungus.
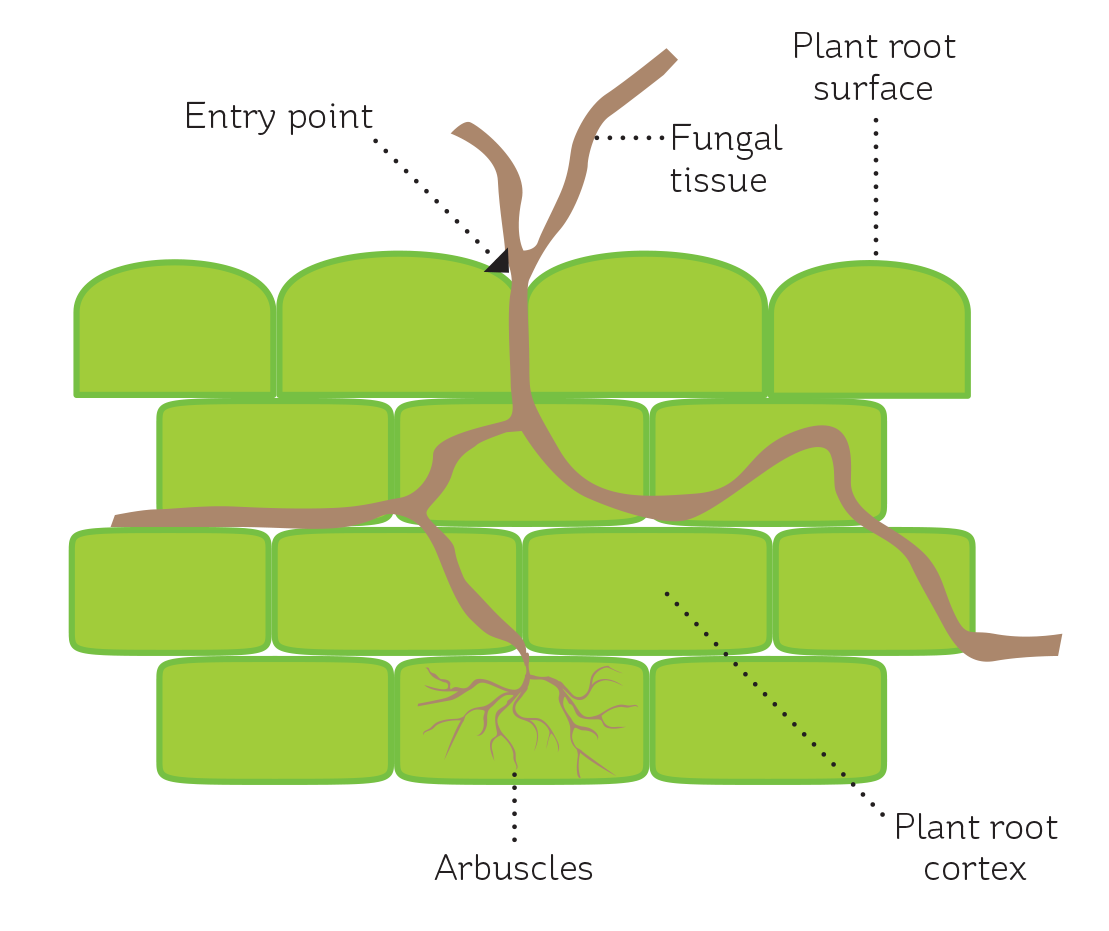
FIGURE 3
A schematic showing plant cell infection by a mycorrhizal fungus
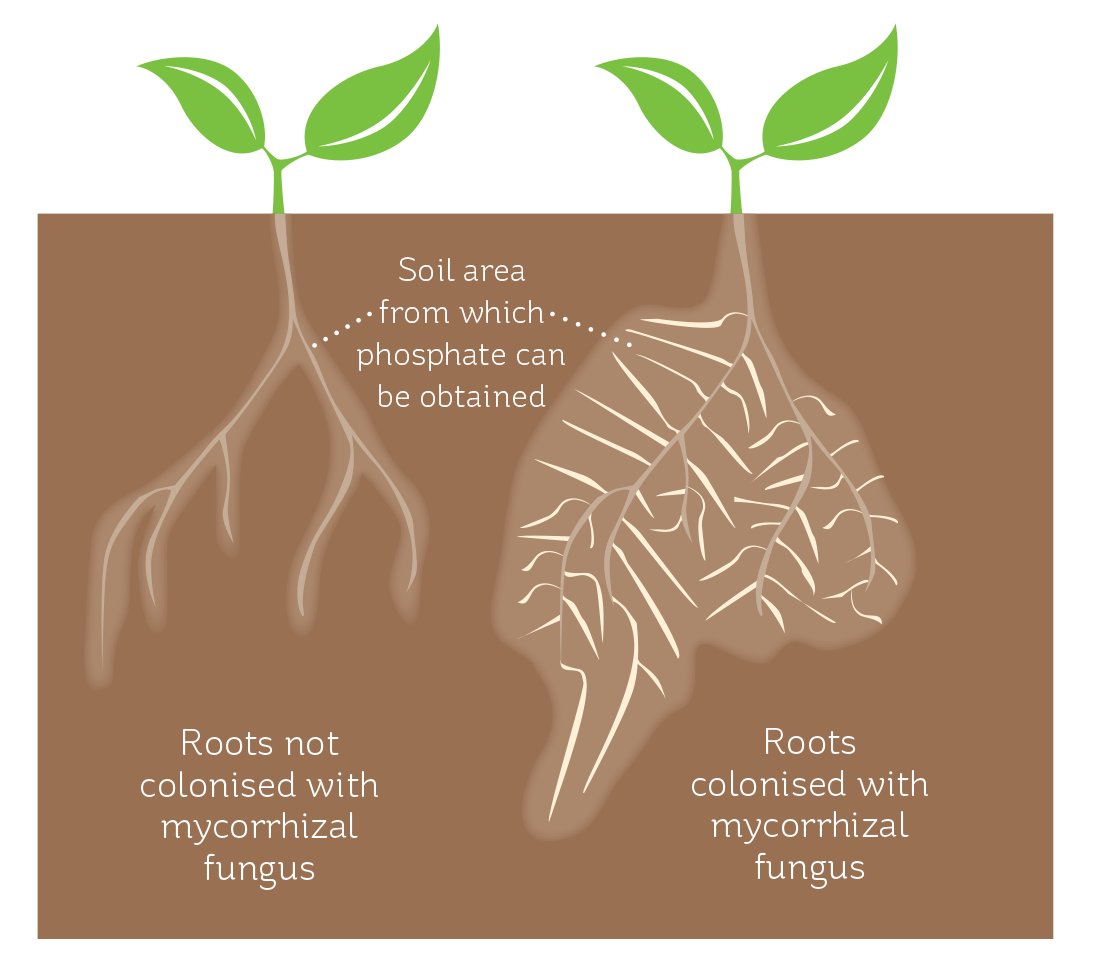
FIGURE 4
Plant roots infected with arbuscular mycorrhizal fungi are able to source phosphate from a greater soil volume.
Read and download the full fact sheet

Understanding phosphorus on your farm
Most New Zealand soils are naturally deficient in phosphorus (P). On farmland, high production of animal and horticultural products exacerbates deficiencies if the phosphorus removed is not replaced. Therefore, the use of phosphorusbased fertilisers is vital for the success of New Zealand agriculture.